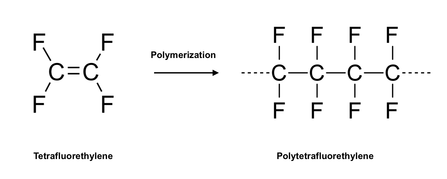
On February 4 of the year 1941, U.S. Patent No. 2,230,654 issued to Roy J. Plunkett for polymerized tetrafluoroethylene, more commonly referred to as polytetrafluoroethylene or its abbreviation PTFE. Still more commonly, PTFE is known by the trademark TEFLON® under which coatings and other polymeric products containing PTFE are sold by The Chemours Company, which was spun off from DuPont, who formerly owned the trademark. As described below, both Plunkett's invention of PTFE and its commercialization occurred by happenstance.
At the time of the invention of PTFE, Plunkett was a DuPont chemist working for the Kinetic Chemical Co., which was a joint venture that DuPont and General Motors had set up to manufacture FREON refrigerant, which had recently been developed to replace the dangerous ammonia and sulfur dioxide that had previously been used. FREON refrigerant was a chlorofluorocarbon (CFC) that had been invented by Thomas Midgley, Jr. and Albert Henne for the Frigidaire division of General Motors. Oddly enough, Plunkett was tasked with developing a refrigerant that could be sold to companies other than Frigidaire, without infringing Frigidaire's patents covering FREON refrigerants.
In his quest for a new refrigerant, Plunkett decided to react tetrafluoroethylene (gas) with hydrochloric acid. In order to do so, Plunkett first had to make a sufficient quantity of tetrafluoroethylene, which he stored in pressurized metal cylinders cooled with dry ice. When the time came to release the tetrafluoroethylene gas from one of the cylinders into a reaction chamber containing hydrochloric acid, nothing came out of the cylinder.
Initially, Plunkett and his assistant were perplexed because they knew from weighing the cylinder that the tetrafluoroethylene had not escaped from the cylinder. Plunkett was also aware of the conventional wisdom that tetrafluoroethylene and other perfluorinated alkenes couldn't polymerize. However, when Plunkett and his assistant cut open the cylinder and found it lined with a slippery white substance, he suspected that the tetrafluoroethylene had indeed polymerized (to form PTFE).
Plunkett's suspicions were confirmed when tests on the white substance determined that it contained fluorine. But that's about all the tests were able to determine. The PTFE was incredibly non-reactive; being insoluble in all known solvents and immune to the strongest acids and bases. In addition, it was difficult to mold and expensive to produce. Nonetheless, Plunkett was able to reproduce the conditions that resulted in the formation of PTFE and describe them in his patent application. Interestingly, claim 1 of Plunkett's patent has to be one of the shortest, if not the shortest, claim of any U.S. patent. Claim 1 consists of only two words: "Polymerized tetrafluoroethylene".
Because of its intractable nature, PTFE probably would have slipped into obscurity as an interesting but worthless byproduct of a failed experiment if not for World War II. The development of the atomic bomb required the separation of U-235 from U-238 using uranium hexafluoride, which was extremely corrosive. The Army was looking for a material that could withstand the corrosiveness of uranium hexafluoride and became interested in PTFE after hearing rumors about its incredible properties. Once testing confirmed the usefulness of PTFE, the Army had DuPont mold PTFE into seals and gaskets for use in handling uranium hexafluoride to produce U-235. PTFE was also found to be transparent to radar, which made it useful for forming nose cones for proximity shells.
The military uses of PTFE were the only uses of PTFE that occurred until after the end of World War II. The success of PTFE in its military applications convinced DuPont to explore commercial use of PTFE. However, it took another 15 years before PTFE began to appear in commercial products on a large-scale due to the difficulty and expense of forming and processing PTFE. Today, PTFE is used in many different products, with non-stick cookware being the most commonly known.
At the time of the invention of PTFE, Plunkett was a DuPont chemist working for the Kinetic Chemical Co., which was a joint venture that DuPont and General Motors had set up to manufacture FREON refrigerant, which had recently been developed to replace the dangerous ammonia and sulfur dioxide that had previously been used. FREON refrigerant was a chlorofluorocarbon (CFC) that had been invented by Thomas Midgley, Jr. and Albert Henne for the Frigidaire division of General Motors. Oddly enough, Plunkett was tasked with developing a refrigerant that could be sold to companies other than Frigidaire, without infringing Frigidaire's patents covering FREON refrigerants.
In his quest for a new refrigerant, Plunkett decided to react tetrafluoroethylene (gas) with hydrochloric acid. In order to do so, Plunkett first had to make a sufficient quantity of tetrafluoroethylene, which he stored in pressurized metal cylinders cooled with dry ice. When the time came to release the tetrafluoroethylene gas from one of the cylinders into a reaction chamber containing hydrochloric acid, nothing came out of the cylinder.
Initially, Plunkett and his assistant were perplexed because they knew from weighing the cylinder that the tetrafluoroethylene had not escaped from the cylinder. Plunkett was also aware of the conventional wisdom that tetrafluoroethylene and other perfluorinated alkenes couldn't polymerize. However, when Plunkett and his assistant cut open the cylinder and found it lined with a slippery white substance, he suspected that the tetrafluoroethylene had indeed polymerized (to form PTFE).
Plunkett's suspicions were confirmed when tests on the white substance determined that it contained fluorine. But that's about all the tests were able to determine. The PTFE was incredibly non-reactive; being insoluble in all known solvents and immune to the strongest acids and bases. In addition, it was difficult to mold and expensive to produce. Nonetheless, Plunkett was able to reproduce the conditions that resulted in the formation of PTFE and describe them in his patent application. Interestingly, claim 1 of Plunkett's patent has to be one of the shortest, if not the shortest, claim of any U.S. patent. Claim 1 consists of only two words: "Polymerized tetrafluoroethylene".
Because of its intractable nature, PTFE probably would have slipped into obscurity as an interesting but worthless byproduct of a failed experiment if not for World War II. The development of the atomic bomb required the separation of U-235 from U-238 using uranium hexafluoride, which was extremely corrosive. The Army was looking for a material that could withstand the corrosiveness of uranium hexafluoride and became interested in PTFE after hearing rumors about its incredible properties. Once testing confirmed the usefulness of PTFE, the Army had DuPont mold PTFE into seals and gaskets for use in handling uranium hexafluoride to produce U-235. PTFE was also found to be transparent to radar, which made it useful for forming nose cones for proximity shells.
The military uses of PTFE were the only uses of PTFE that occurred until after the end of World War II. The success of PTFE in its military applications convinced DuPont to explore commercial use of PTFE. However, it took another 15 years before PTFE began to appear in commercial products on a large-scale due to the difficulty and expense of forming and processing PTFE. Today, PTFE is used in many different products, with non-stick cookware being the most commonly known.
Proudly powered by Weebly